Freeze Thaw or Cyclic Temperature stress tests are designed on the knowledge of the product so as to mimic likely storage conditions. The period of cycle is mostly considered 24 hours since the diurnal rhythm on earth is 24 hours.
Additionally, the distribution environment can vary greatly, especially when a drug product is shipped between various climatic zones. Seasonal changes, mode of transportation, and the number of drop-off points are also variables that should be considered within the pharmaceutical supply chain. Drug products requiring controlled-temperature storage conditions must be distributed in a manner that ensures that the product quality will not be adversely affected. With the exception of short transit times within the same climatic zone, it is virtually impossible to validate a shipping method against all environmental conditions.
Therefore, distribution stability studies are designed to generate additional data to complement preformulation development and routine (ICH) Q1A(R2).
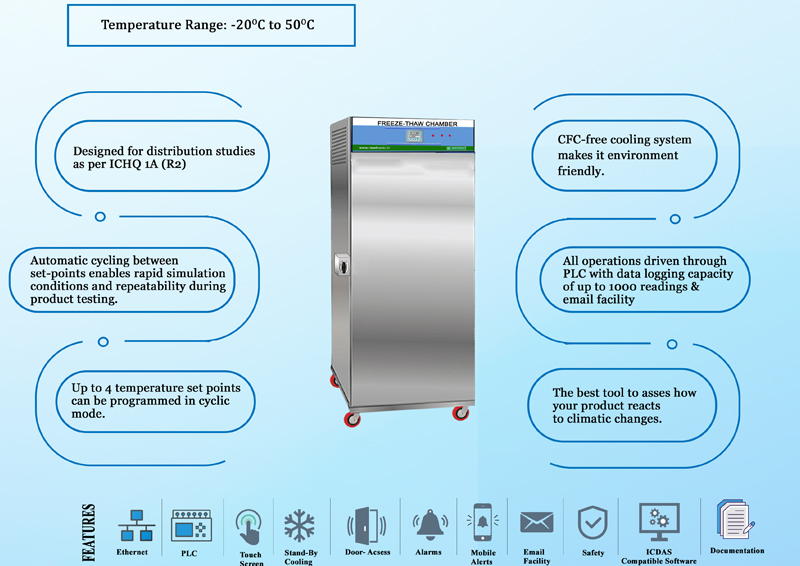
NEWTRONIC Freeze Thaw Chambers control a broad temperature range of -20°C to 60°C thereby offering the flexibility to simulate a broad range of conditions. The min temperature, max temperature & cyclic time are all user-settlable thereby allowing the user to program a specific profile for their studies. Automatic cycling between the setpoints enables rapid simulation of conditions & the need to replicate freezing and thawing during product testing.

Stability (Reach In) Chamber

Stability ( Walk-In) Chamber

Freeze Thaw Chamber

Photostability Chamber

Ecotronic Chamber